 For instance, the first element in the periodic table, hydrogen, has an atomic number of 1, because a hydrogen atom has only 1 proton. At the other extreme, an atom of the synthetic element oganesson has 118 protons and an atomic number of 118. The elements are also organized into groups, or families, that share similar physical and chemical properties. Each column in the periodic table, going from top to bottom, represents one of these groups. Additionally, related elements are usually color-coded to indicate whether they are metals, semi-metals, or non-metals. When you read the period table in rows going across from left to right, you will find that the elements in each row share the same number of atomic orbitals. The orbitals are the regions around the atom’s nucleus where electrons are most likely to appear. Each of these 7 rows is called a “period.” Finally, you might notice that there are gaps in the periodic table, particularly in the top 3 rows. This is because the elements don’t always fit neatly into groups that share the same physical and chemical characteristics. One group of elements that have nearly indistinguishable chemical properties, called the rare earth elements, is separated out into its own separate space at the bottom of the periodic table. 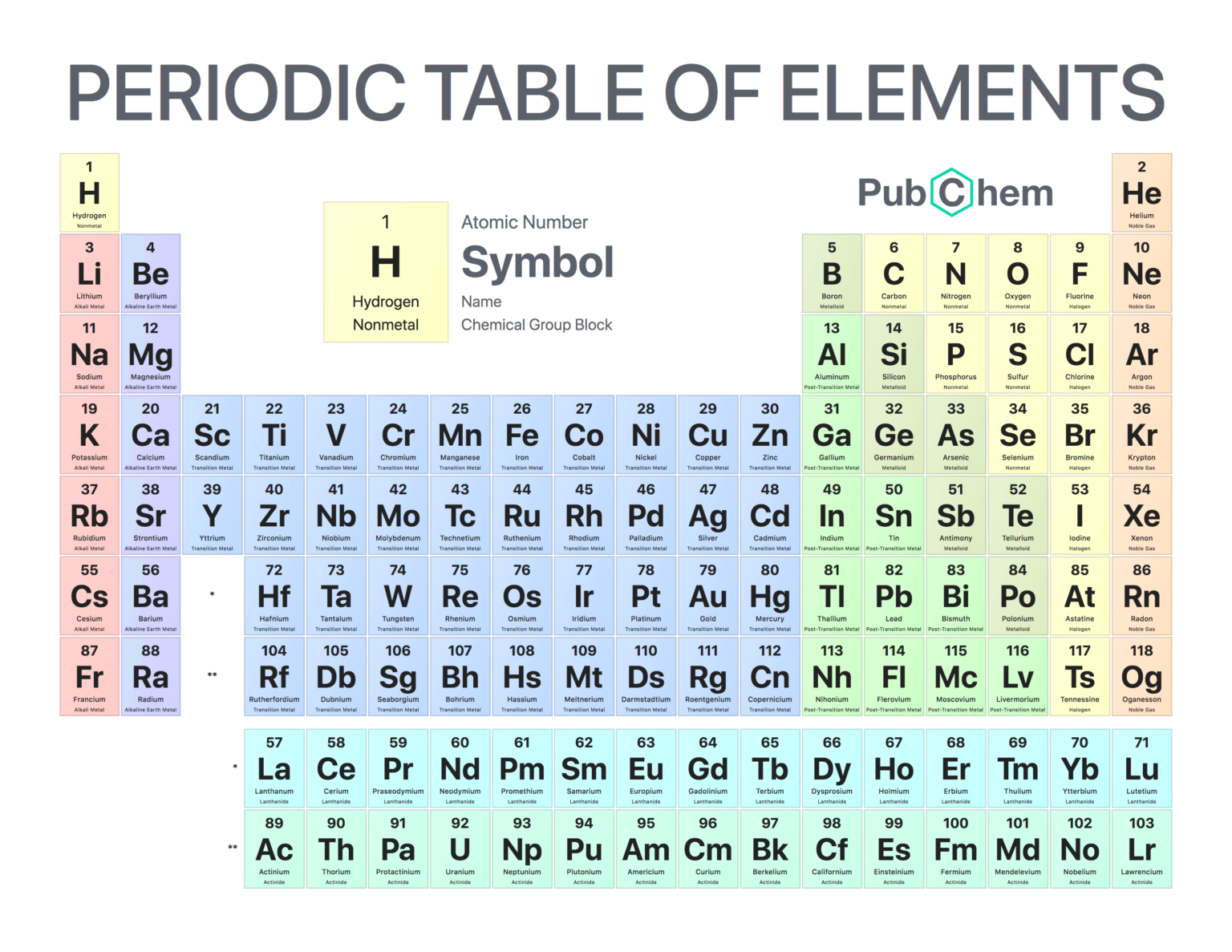
When you look at individual elements on the periodic table, there are several pieces of information that you can find on each square. For example, every element has a 1 to 3-letter symbol that represents the name of the element. While some of these are pretty obvious, like O for oxygen and C for carbon, others don’t necessarily match the common name of the element. For instance, the symbol for lead is Pb, from the Latin word for lead, plumbum. In addition to the element symbol, you’ll also see the atomic number at the top of the square, which represents the number of protons in an atom of the element. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
